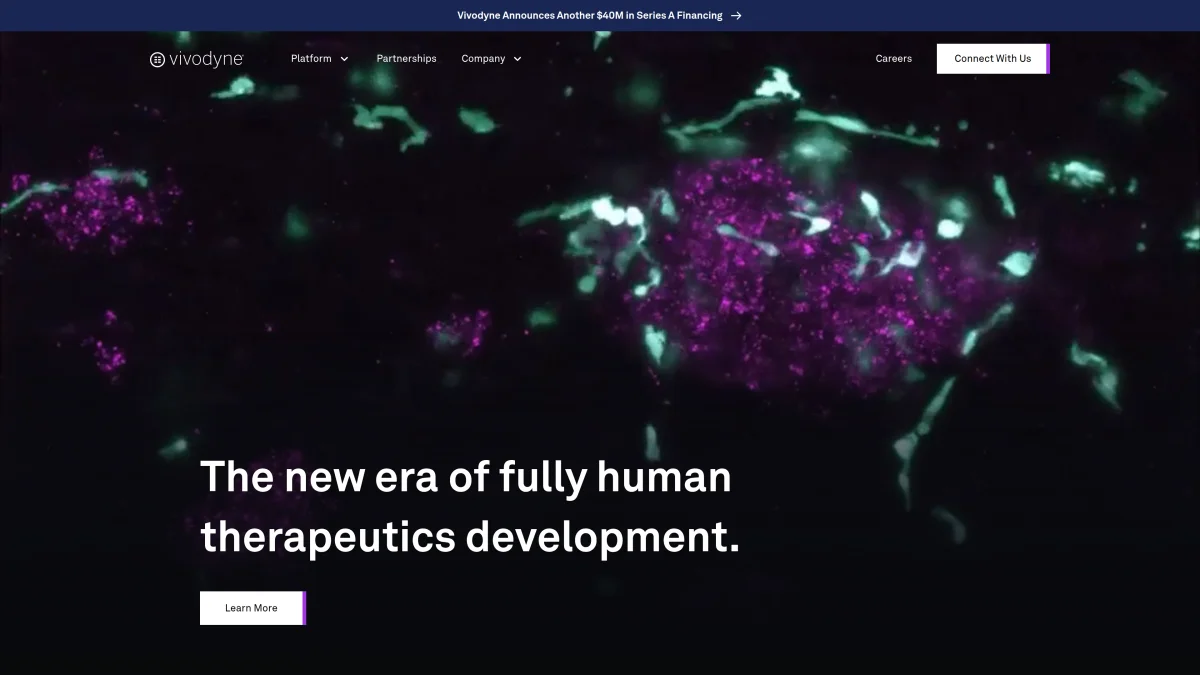
Vivodyne, a biotechnology company pioneering a new approach to drug discovery, has secured $40 million in Series funding. The round was led by Khosla Ventures, with participation from Lingotto Investment Management, Helena Capital, Fortius Ventures, and existing investors Kairos Ventures, CS Ventures, Bison Ventures, and MBX Capital. This significant investment will fuel Vivodyne's mission to replace unreliable animal testing with a more accurate and efficient human-tissue-based model.
Vivodyne's innovative technology utilizes automated robotic platforms and AI to grow and analyze thousands of fully functional human tissues. This allows pharmaceutical companies to obtain clinically relevant data at a massive scale, significantly reducing the risk of clinical trial failures (currently estimated at 95%). The company's platform is already used by most leading pharmaceutical companies.