AI is no longer confined to digital realms. In a significant leap for biology, GPT-5 has been directly linked to an automated laboratory, demonstrating a 40% reduction in the cost of producing proteins through cell-free synthesis. This collaboration with Ginkgo Bioworks showcases how frontier AI models can now drive physical experimentation at scale.
AI Powers Wet Lab Revolution
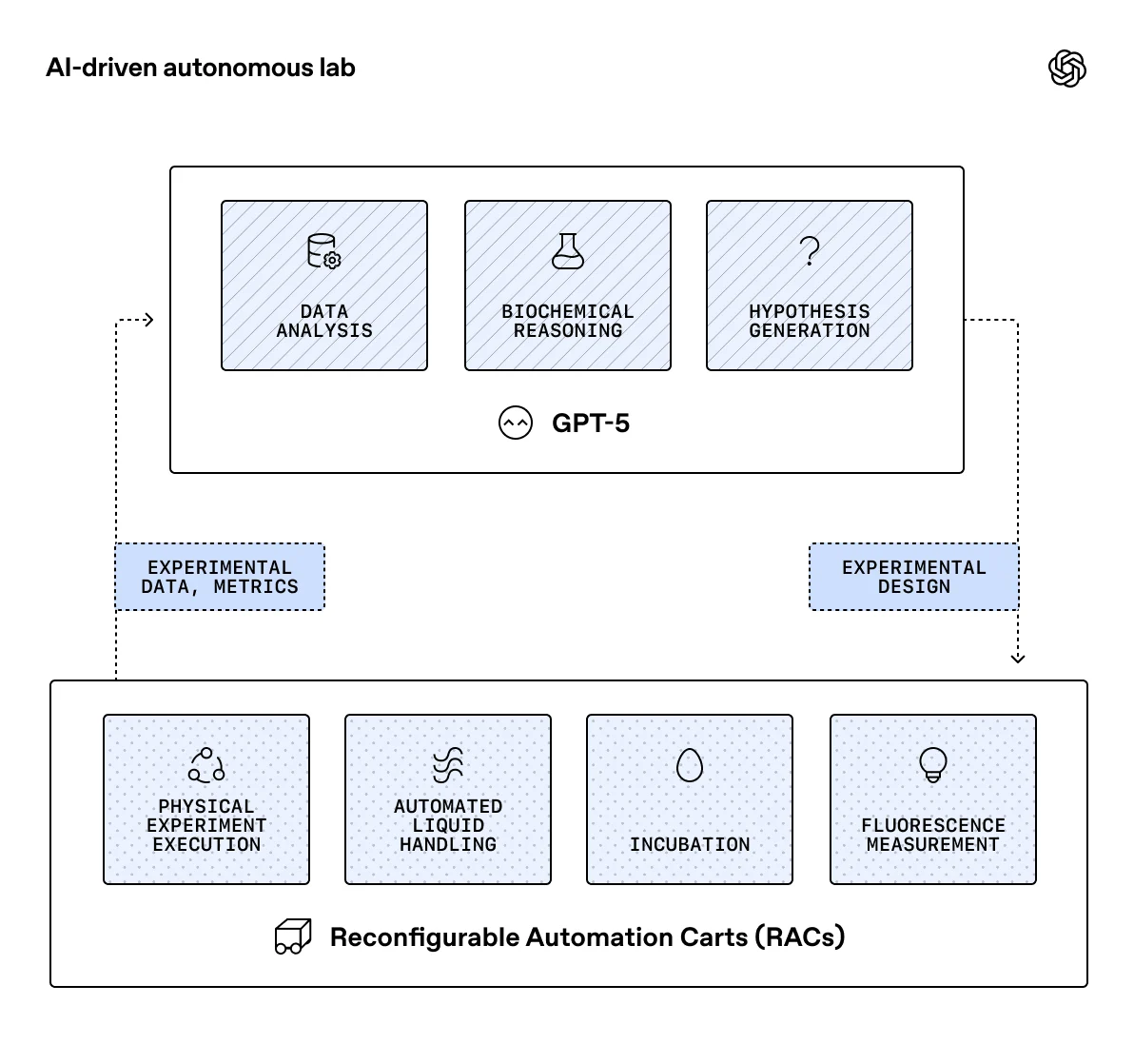
Unlike fields where ideas can be tested computationally, biological progress traditionally relies on time-consuming and expensive lab work. Autonomous labs, however, are changing this paradigm. By connecting advanced AI models like GPT-5 to robotic systems, researchers can now propose experiments, execute them, analyze results, and iterate autonomously, drastically reducing experimental bottlenecks.
Building on prior work that showed GPT-5 could optimize lab protocols, this new research focuses on a critical biological process: cell-free protein synthesis (CFPS). This method allows for protein production without living cells, making it ideal for rapid prototyping and testing.
Optimizing Protein Production
The partnership involved linking GPT-5 to Ginkgo's cloud laboratory, a remotely operated, automated wet lab. Over six rounds of closed-loop experimentation, the system tested over 36,000 unique CFPS reaction compositions. GPT-5, given access to computational tools and relevant scientific literature, rapidly established a new cost-efficiency benchmark.
The system achieved a 40% reduction in overall protein production cost and a 57% decrease in reagent costs. Crucially, it also identified novel reaction compositions that proved more robust under the conditions typical of automated labs.