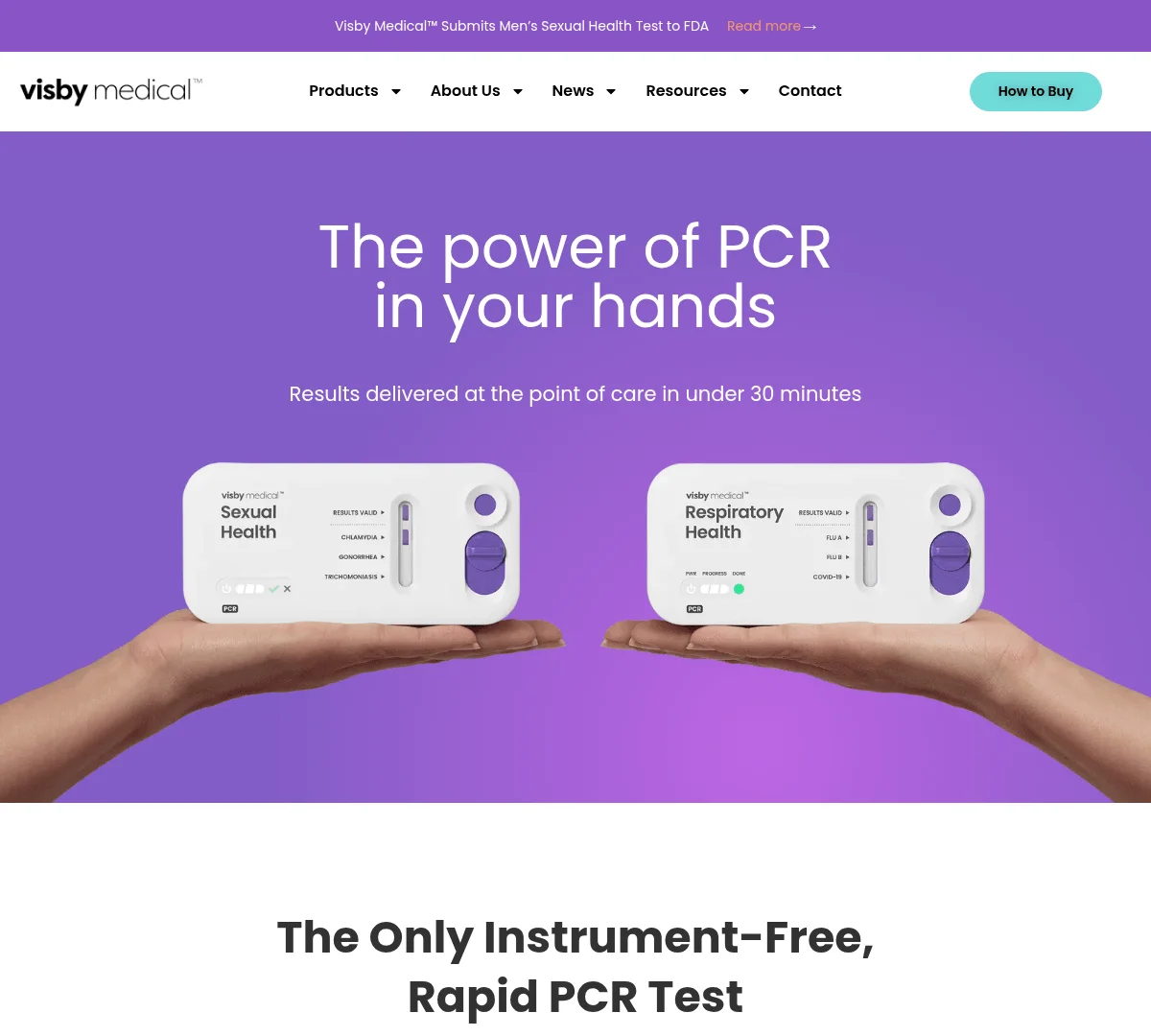
Visby Medical has raised up to $65 million in a round led by Catalio Capital Management to launch the first FDA-authorized at-home PCR test for sexually transmitted infections (STIs). The company’s Women’s Sexual Health Test detects and differentiates three major infections—Chlamydia, Gonorrhea, and Trichomoniasis—with lab-grade accuracy in under 30 minutes, using a single-use, instrument-free PCR device.
Unlike traditional diagnostics that require lab equipment and processing delays, Visby Medical’s technology integrates automated real-time PCR (RT-PCR) into a compact, disposable format operable via a smartphone app. Positive results trigger an automatic referral to a telemedicine provider, streamlining diagnosis, consultation, and treatment.